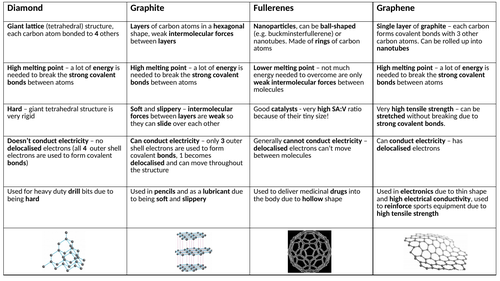
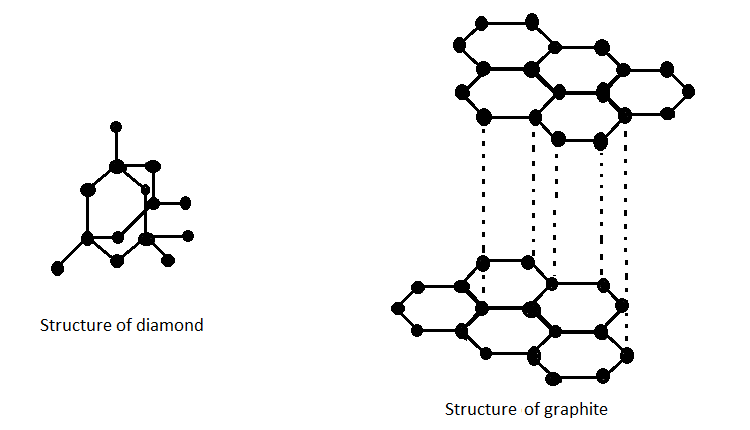
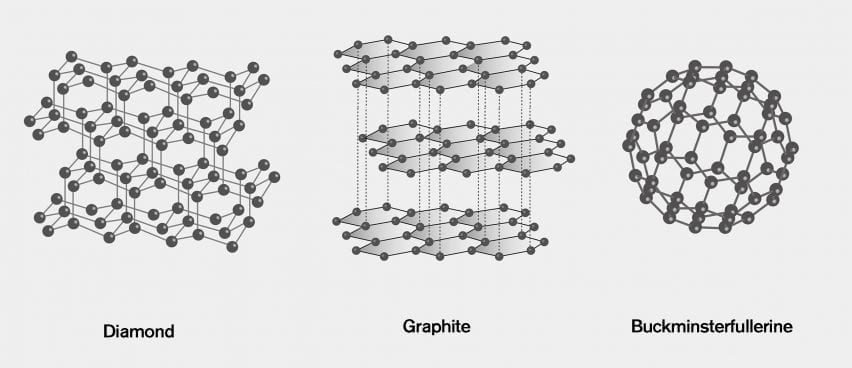
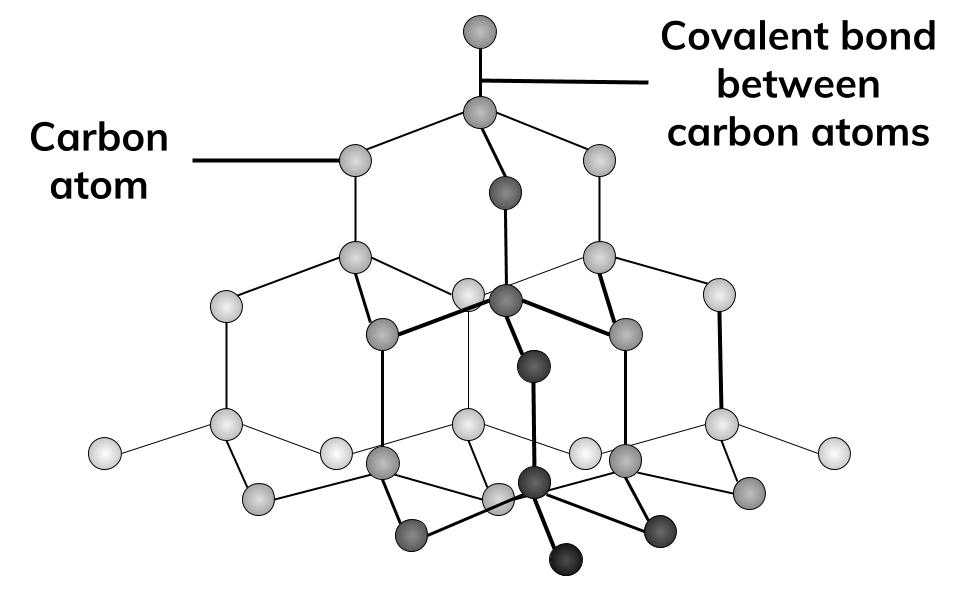
Draw the structure of graphite and diamond. Explain how the structures are responsible for the difference in their physical properties.

Carbon Allotropes Graphite, Diamond, Fullerene Atomic Structures. Stock Vector - Illustration of element, diagram: 192490784
1:50 explain how the structures of diamond, graphite and C60 fullerene influence their physical properties, including electrical conductivity and hardness - TutorMyself Chemistry
Giant covalent structures - Structures and properties - GCSE Chemistry (Single Science) Revision - WJEC - BBC Bitesize

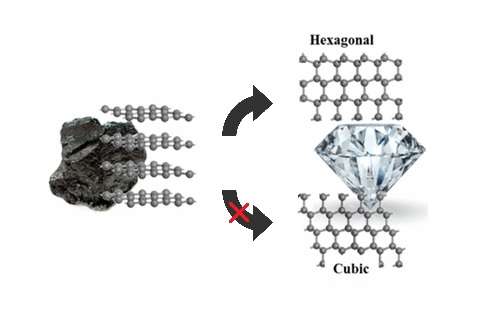
The transformation of diamond to graphite: Experiments reveal the presence of an intermediate linear carbon phase - ScienceDirect

How can graphite and diamond be so different if they are both composed of pure carbon? | Scientific American

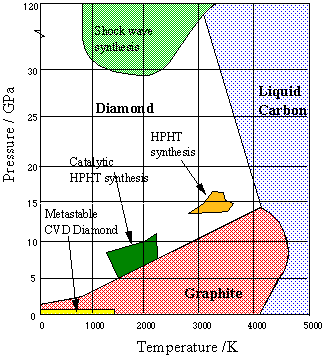
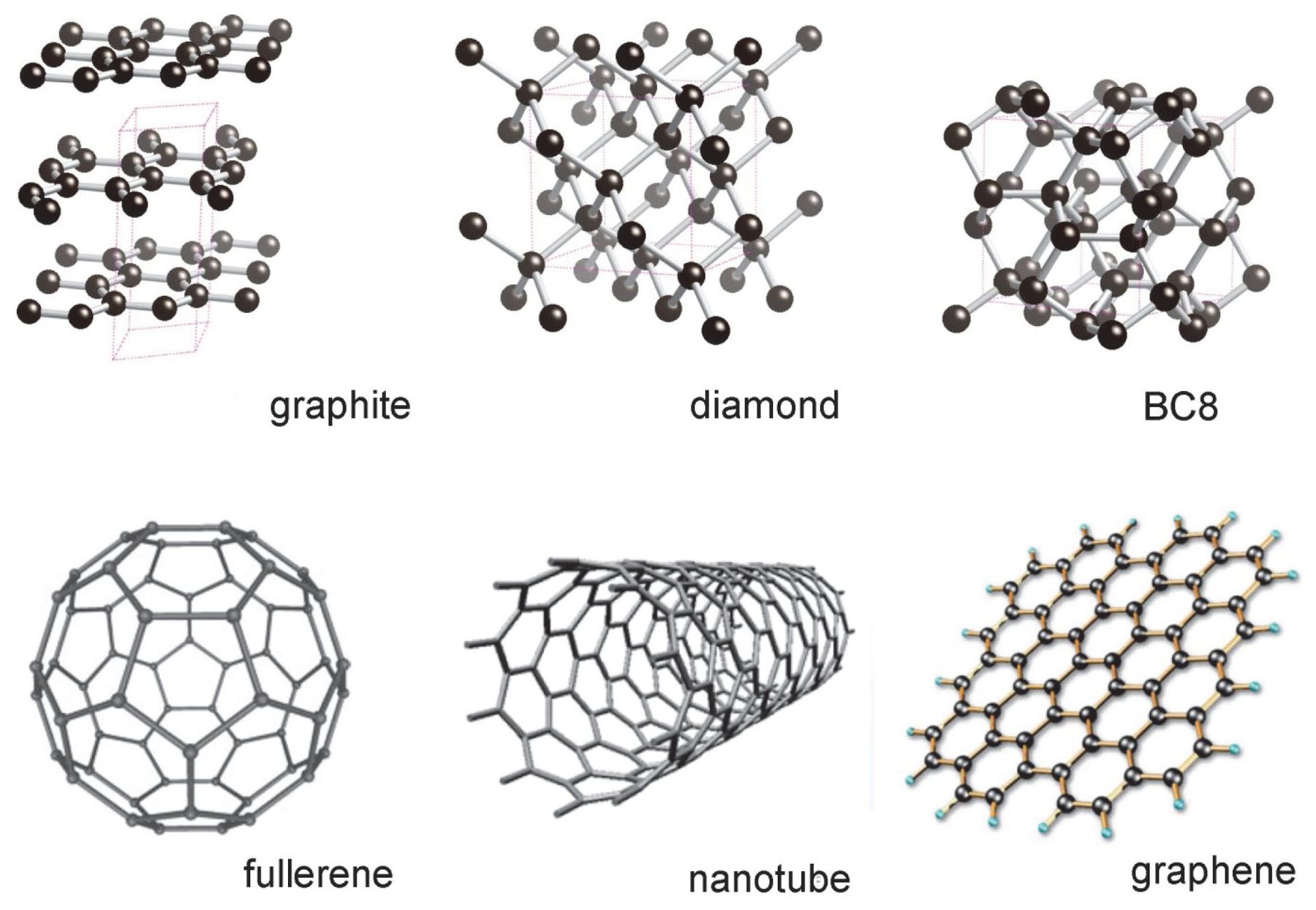






![Class 10] What do you mean by Allotropes of Carbon? - Teachoo Class 10] What do you mean by Allotropes of Carbon? - Teachoo](https://d1avenlh0i1xmr.cloudfront.net/0b33a27a-2f40-4457-a170-fdfc5d80dd4f/allotropes-of-carbon---teachoo.jpg)